Bio-Standards’ Biocompatibility Evaluation Solution is designed to guide medical device manufacturers through the essential process of demonstrating biocompatibility, ensuring adherence to stringent regulatory standards. Our approach includes a series of tailored steps to comprehensively address biological safety assessments:
Biocompatibility Documentation Process
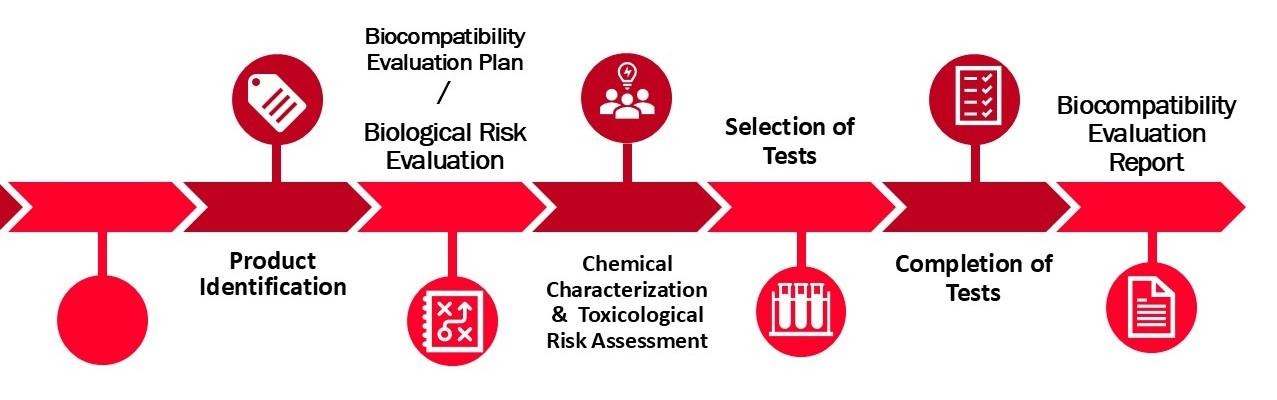
Initial Product Identification Meeting: This vital first step involves in-depth consultation with manufacturers and our biocompatibility experts to understand the product's characteristics, intended use, and interactions with the human body. This discussion helps define the scope of necessary biocompatibility assessments.
Comprehensive Biological Risk Assessment: We perform a thorough biological risk assessment to identify potential hazards associated with the product's materials and intended use. This assessment forms the foundation for a focused biocompatibility evaluation strategy.
Biocompatibility Evaluation Plan Preparation: Utilizing insights from the risk assessment, we create a Biocompatibility Evaluation Plan that outlines the specific tests and evaluations needed to assess biocompatibility, considering the nature and duration of body contact.
Guidance on Test Facility Selection: Following the establishment of the Evaluation Plan, our experts assist in selecting the appropriate testing facilities, ensuring all tests meet regulatory standards and industry best practices.
Biocompatibility Evaluation Report Compilation: Once testing is complete, we prepare the Biocompatibility Evaluation Report, consolidating test results and providing a detailed analysis of the product’s biocompatibility and compliance with applicable regulations.
Comprehensive Support Offered
Our solution includes extensive support throughout the process:
At Bio-Standards, we are committed to assisting medical device manufacturers through the complexities of biocompatibility assessments, from initial evaluations to final reporting. Our objective is to ensure your product meets all necessary safety and regulatory standards, facilitating successful market entry and prioritizing patient safety. Contact us to discover how our Biocompatibility Documentation Solution can support your compliance journey.
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray

