Bio-Standards’ Risk Management Solutions are expertly designed to enhance product safety and ensure compliance with SFDA regulations and internal standards ISO 14791. Our collaborative approach with medical device companies and experts ensures thorough risk mitigation and product integrity through the following structured steps:
Product Identification Session: Conduct a comprehensive analysis of the product’s characteristics, intended use, and potential risks. This collaborative session involves both medical device companies and our specialists to ensure a clear understanding of the product landscape.
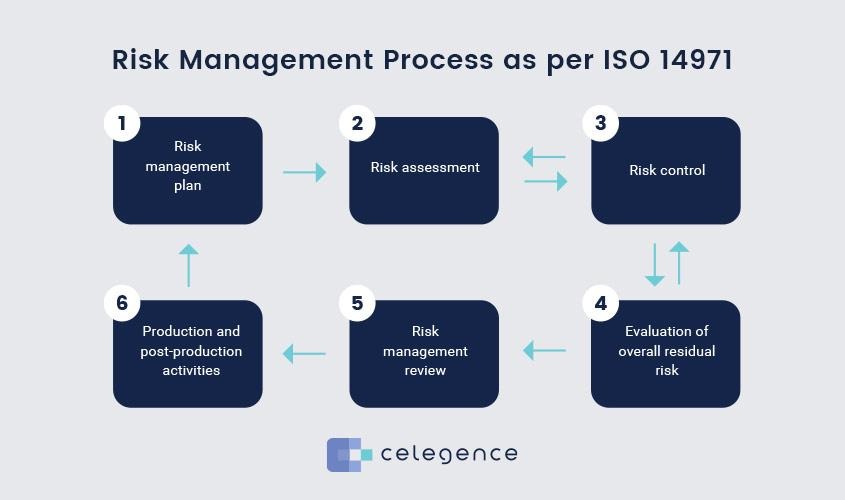
Risk Management Plan Development: Develop a customized risk management plan following a detailed product analysis. This plan covers risk identification, analysis, control measures, and regular reviews to maintain proactive risk management.
Comprehensive Risk Analysis Documentation Preparation: Facilitate an exhaustive risk analysis document based on the risk management plan, detailing identified risks and proposed mitigation strategies. This document serves as a blueprint for risk control.
Risk Management Report Compilation: Formulate a comprehensive report detailing identified risks, implemented measures, and the strategy’s effectiveness, offering transparency throughout the process and discuss any further required actions for continued risk mitigation.
At Bio-Standards, we support your path through the complexities of risk management. Our adaptable solutions provide expert guidance, whether you’re initiating a risk management plan, conducting a risk analysis, or completing a risk review. Our goal is to minimize risks prior to market launch and ensure compliance with all regulatory standards, standing with you at every step to ensure your products meet the highest safety benchmarks.
With proven experience supporting 1,600+ Medical Device manufacturers and 120,000+ product registrations, Bio-Standards ensures your validation and stability studies meet the highest regulatory standards.
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray

