Navigating the complex regulations of medical device compliance with the SFDA is critical for market success. At Bio-Standards, we specialize in preparing regulatory application files, ensuring your device's compliance journey is seamless. Our experts create meticulously tailored files, in a comprehensive or section-specific format, to meet your unique needs.
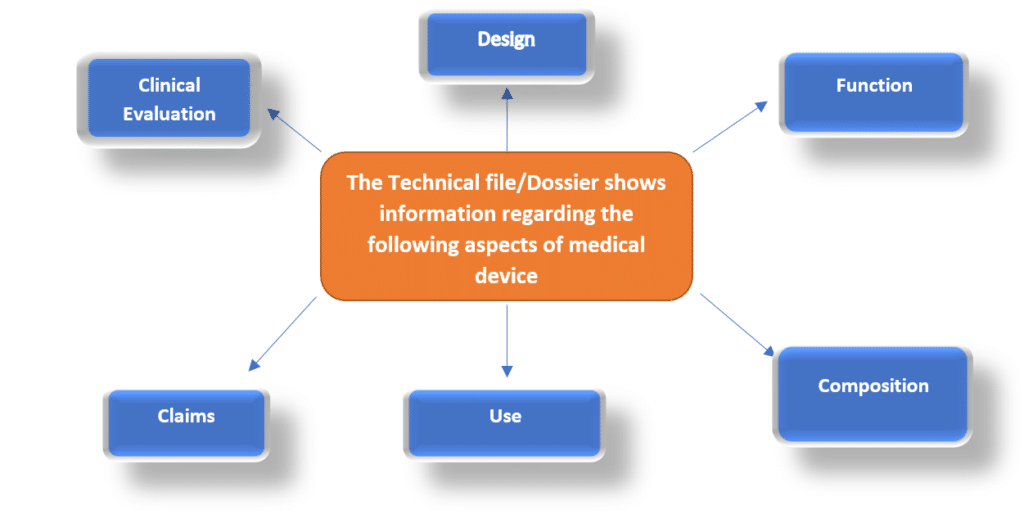
Technical File Structure: A Compliance Blueprint
Our support extends to usability documentation, PMS plans, and process validations, ensuring comprehensive compliance documentation.
Our holistic approach to regulatory file preparation not only facilitates approval but also lays a solid foundation for successful market entry and long-term compliance. Bio-Standards is committed to guiding you through this intricate process with precision and efficiency.
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray
Mindray
Acon Biotech (Hangzhou) Co.,Ltd.
Integra LifeSciences Services
Semac for medical services
ALMADAR MEDICAL Co.
CenteMed Medical Establishment
Al-Kawader Scientific Company for Trading & Medical Supplies
Al-Kawader Scientific Company for Trading & Medical Supplies
Bgi Genomics Co., Ltd
KANEKA PHARMA EUROPE NV
Meditek Trading Est
Allengers
Medical ActTect
Keydent est
MicroSafe
Lexington Medical, Inc.
Julwan For Trading ESt.
Inbentus Medical Technology
Koning International
RMDM
National Excellence for Business Solutions (NEXCO)
Maxigen Biotech Inc.
EIZO Corporation
Medprin Biotech GmbH
Elipse Health Limited
Huawei
Inbentus Medical Technology (Spain)
Centennial Medical Establishment
Elipse Health Limited (United Kingdom)
ALMADAR MEDICAL
KANEKA PHARMA EUROPE NV
Allengers
NingboSoundway Medical Appliances Co , Ltd
SHENZHEN MINDRAY BIO-MEDICAL ELECTRONICS CO., LTD.
PMLS Upstream Marketing Department of Mindray

